Biopharmaceutical Manufacturing: An Evolving Industry
David Gemmell discusses how chemical engineers play their part in modern-day biopharma
IN THE second article of our biopharmaceutical manufacturing series, we explore how chemical engineers are involved in the manufacture of different products from traditional vaccines to more cutting-edge modalities like mRNA-based therapeutics. We will also discuss some of the modern manufacturing philosophies striving to become routine in an industry so resistant to risk and change.
In the late 19th Century, Europe and America were experiencing major outbreaks of poliovirus. By 1952 polio had become one of the leading causes of death for children in the US, with an estimated 20,000-35,0001 people dying or being paralysed each year, with case numbers rising among young adults.
In 1956 an alarming number of teenagers were being paralysed by the virus, despite there being a widely-available vaccine that had been demonstrated to significantly increase one’s immunity.
Young Americans had not been suitably persuaded to receive the vaccine which would increase the immunity of the overall population. Interest in the vaccine changed on 28 October 1956, when a young Elvis Presley was vaccinated live on The Ed Sullivan Show. Within six months the vaccination rate in teens in the US soared.
This was a watershed moment in 20th Century vaccine history, and it raised interesting perspectives on how to effectively communicate and engage with the public. Whether Elvis’ live immunisation contributed significantly to the increased uptake, or whether it was the continued rollout of medical information and vaccine advocacy in society, is up for debate. Either way, the real credit should go to Jonas Salk2 for developing the vaccine in the first place. In the modern era, thankfully, poliovirus has been almost eliminated from the global population.
Let’s take a look at what the King of Rock and Roll was injected with, and how it differs from the vaccines many of us have received during the Covid-19 pandemic.

Common vaccine manufacturing
All vaccines are designed to provide your body with the tools necessary to minimise the impact of an infection or to prevent infection altogether. There are many different types of vaccines, each with a unique manufacturing approach.
The polio vaccine consists of an inactivated version of the polio virus itself2, a rudimentary type of vaccine which gives the immune system the ability to recognise and destroy infectious agents after coming into contact with a “dead” version of the virus. The inactivated virus cannot hijack the cellular machinery in the body to replicate, propagate and cause further harm.
Broadly speaking, inactivated virus vaccines are considered safer3 than an attenuated virus vaccine, which utilises a weakened form of the “live” or more accurately a “replication competent” virus (viruses are generally not considered to be truly alive). In rare circumstances, attenuated viruses can mutate and begin to replicate within the host and cause harm. However, inactivated virus vaccines typically offer a shorter course of protection3 than an attenuated virus and this must be considered during development.
One of the most common vaccines manufactured in the UK is the influenza vaccine. This inactivated virus vaccine is typically produced using an egg-based process. The “live” influenza virus is injected into chicken eggs, the virus grows and replicates within the egg; the virus, which has replicated to a high concentration, is inactivated, separated, and purified. While the concept of millions of chicken eggs being used to manufacture the seasonal flu vaccine does not sound like the most cutting-edge biopharmaceutical process, it is a tried-and-tested methodology for making a critical vaccine that is distributed on a large scale prior to every flu season.
Unlike other high-value biologics on the market, unit cost is more of a major aspect for vaccine manufacturers to consider. Vaccines which have a higher unit cost are less likely to be adopted for wide scale use and will have limited markets in the developing world. Manufacturers using egg-based production methods keep costs low, but this approach is not without inherent challenges.
Variable virus growth within the eggs leading to inconsistent yield and the potential for avian influenza strains contaminating the process must be accounted for. There are also difficulties associated with virus separation, purification and post-process cleaning of sticky collagen and albumen proteins contained within eggs.
Chemical engineers must design, install, commission, and validate thorough cleaning and sterilisation processes to eliminate the risk of batch-to-batch contamination. Mixing steps, transfer piping, tangential flow filtration (TFF) systems, and filter housings must be designed to be inherently hygienic. Ensuring high surface finishing, free-drainable systems and careful process cleaning parameters allow product quality to remain high. High quality water, typically produced by reverse osmosis or distillation, is used either for cleaning or in the product itself. These systems must be sized to eliminate process bottlenecks.
To eliminate bacterial growth, water in storage vessels must not be allowed to stagnate. Strict requirements and specifications for loop circulation systems, in-line velocity, Reynolds number and minimum temperature all help to mitigate the growth of unwanted bacteria and biofilms within the manufacturing facilities’ utility or production systems.
New ways of doing old things
Companies are assessing whether the use of cell-based systems can replace eggs for vaccine production. Adherent or suspension cell culture systems (where the cells grow in monolayers adhered to a surface or are suspended in liquid) for influenza can have typical process yields of 35%.4 One litre of the product can provide between 50 and 100 vaccines, with many of the legacy processing challenges removed. However, the legacy egg approach is a well characterised process which has been used for 30 years, it’s cheap and effective – leaving an interesting decision point for manufacturers to consider.
One of the most topical advancements in the biotherapeutic landscape has been the rapid development and global rollout of novel vaccine modalities to combat the Covid-19 pandemic
One of the most topical advancements in the biotherapeutic landscape has been the rapid development and global rollout of novel vaccine modalities to combat the Covid-19 pandemic. It is difficult to find an equivalent example of how such a niche piece of biotechnology became so widely integrated into the public lexicon in such a short space of time. No matter what your occupation, age or where you live, you are likely to have heard of, discussed, and possibly even been immunised by a messenger ribonucleic acid (mRNA) based vaccine.
mRNA based vaccines contain a small strand of genetic information which contains the recipe for a piece of viral protein. Once inside the body’s cells, the mRNA will encounter the machinery to read this recipe and create a string of amino acids which are then modified to become the viral protein, most frequently the virus’ capsid protein– the spike S1 protein from SARS-CoV-2 virus is a pertinent example. Harmless in itself, the presence of capsid proteins elicits production of antibodies, providing protection from a subsequent infection by the whole virus.
While scientists have been working on using mRNA as a therapeutic product for decades, there have been many hurdles to overcome. Historically, it was challenging to facilitate the effective diffusion of mRNA into cells. Additionally, mRNA is delicate and easily digested by enzymes within cells, so even once it enters, it could be destroyed before it has time to act. These challenges have been overcome by improvement in enzymatic capping of the molecule and by wrapping the mRNA in a combination of lipid particles (or other drug delivery systems) which facilitate diffusion into cells and increase the stability of the molecule.
These developments have allowed us to battle the coronavirus pandemic and now industry has its eyes set on using mRNA to address a variety of diseases. As the body translates the mRNA into whichever protein is coded into its genetic sequence, there are many possibilities on the horizon. The industry wants to deliver life-enhancing drugs to a wider population, and that means more types of therapeutics, more flexible manufacturing, decreased cycle times, adoption of platform manufacturing approaches to increase quality, and ultimately a reduction in the cost of goods (COGs).
Evolution in manufacturing
While the influenza vaccine does not typically use a cell-based process, many biopharmaceutical products are made by cells or bacteria in bioreactors/fermenters (see An Introduction to the Biopharmaceutical Industry, TCE 970, for an overview of the monoclonal antibody manufacturing process and other common biopharmaceutical products).
The average titres (concentrations) of biological drug products produced by cells in a bioreactor have increased significantly. Fifteen to twenty years ago, it would have been common to see cell lines and manufacturing processes producing 0.5-1 g5 of drug product per litre of bioreactor volume. Bioreactor size was expected to be the bottleneck in the manufacturing process. Now it is commonly around 5-10 g/L6 for the same products. Bioreactor design, feeding strategies and our understanding of cell culture has facilitated these advances.
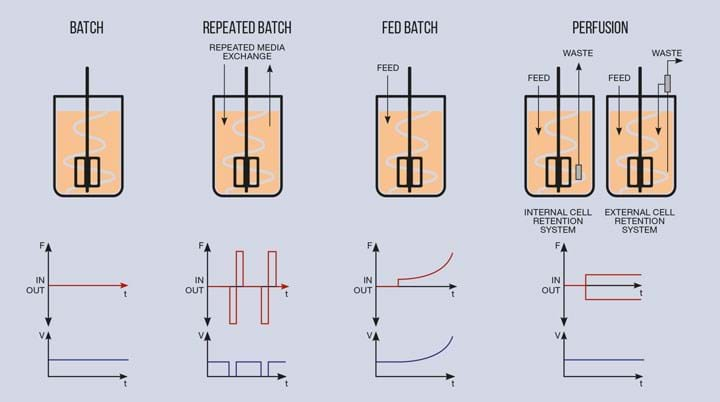
Bioreactor feeding strategies have moved away from traditional “batch”, which involved loading all the necessary cells and nutrients together and leaving it to run its course. Fed-batch (where fresh nutrients are periodically added) and perfusion (where fresh nutrients are added and waste materials removed) allow much higher viable cell numbers and therefore much higher product titres per litre of bioreactor volume. This allows a significantly smaller bioreactor to produce the product previously made in a larger system.
This is a fantastic development from a capital cost perspective. The days of building 15–20,000 L stainless bioreactors with huge steel centrifuges for clarification and all the subsequent large-scale downstream purification hardware and associated cleaning or sterilisation systems are possibly coming to an end. However, higher cell counts, products and waste materials all significantly impact the downstream purification trains, which must be designed to accommodate the extra material.
This leaves many companies and their biochemical/biomanufacturing engineers all pondering the same question. How best to effectively capture the benefits of these modern technologies, novel modalities, and manufacturing approaches?
Single-use systems - typically gamma radiation sterilised plastic bags/tubing/filters -allow efficient turnaround. Their adoption allows manufacturers to pivot quickly, making many different products in a flexible facility that is significantly cheaper and faster to build. But it leaves them exposed to complex supply chains to provide all the irradiated plastic systems needed to manufacture and transfer the delicate biologic through the various batch processing unit operations. These systems are problematic from an environmental waste perspective. However, few industries could justify the usage as well as the biopharma industry. Single-use technology mitigates the risk of any product cross contamination or deviation from system cleaning efficacy.
Established manufacturing sites with large stainless steel systems will still be used for commercialised molecules already on the market. Legacy manufacturers are looking at how to optimise their processes to fend off new competitors on the horizon who are producing “biosimilars”, which are comparable but distinct molecules targeted at existing markets in the same way generics are for small molecule therapeutics.
There is a growing need for chemical engineers to assess existing processes, debottleneck or integrate newer technologies into the established unit operations. This is a daunting task in such a highly regulated industry
Companies that rest on their laurels as patent expiration dates approach may well find that they can’t compete, so there is a growing need for chemical engineers to assess existing processes, debottleneck or integrate newer technologies into the established unit operations. This is a daunting task in such a highly regulated industry as you can’t simply swap out one piece of equipment for another, even if it has an obvious benefit.
Quality and regulatory governance will demand that thorough assessments are undertaken to ensure that no unexpected consequences occur. This process is laborious and expensive but vital in an industry where product quality is the paramount concern. On many occasions even the most obvious process improvements will be rejected because there is no bandwidth to support the complex re-submission to the US FDA (Food & Drug Administration), UK MHRA (Medicine Healthcare Regulatory Agency) or other governing body. Regulatory agencies do not care about process economics, only product quality.
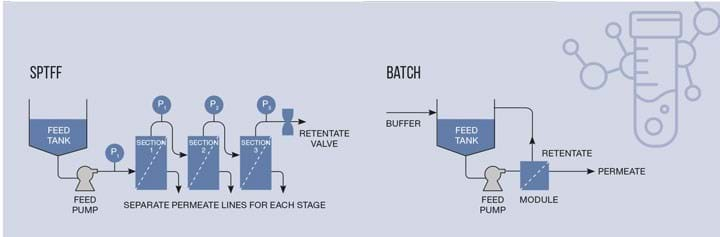
If the cost benefit is positive, and the desire is strong enough, change does occur. Many companies are now using hybrid manufacturing processes, stainless steel and plastic systems striving for a best-of-both-worlds methodology. Modern unit operations like single-pass tangential flow filtration (SPTFF) technologies are being integrated into existing TFF processes to further concentrate drug substance to minimise unit operation size downstream or to achieve more concentrated final products for patients.
Biologics are commonly administered via an intravenous drip, requiring the patient to stay in a hospital for several hours as the dilute drug enters their system. Many companies and clinicians are looking at subcutaneous injection of more concentrated doses that the patient can administer themselves at home. SPTFF is a useful technology to achieve this increased concentration, which can be used after initial concentration using a traditional TFF step or as a direct alternative to standard TFF.
SPTFF does not require the product to be recirculated around the system over and over as the concentration occurs, it simply enters into the filter membranes which are arranged in series. This can be implemented by modifying existing TFF systems with relatively limited costs and many benefits, or by purchasing single-use capsules for even faster turnarounds.
Designing stainless systems for quick turnaround novel modalities can be problematic, and with batch sizes often less than 1,000 L this approach may not be as effective as utilising single-use manufacturing systems to capture those much sought-after COG reductions.
Smaller manufacturers using modern processing techniques with single-use systems and high titre upstream operations are becoming much more prevalent. Increased efficiency in upstream production must be balanced with a similar increase in downstream purification efficiency. Adapting existing systems and undertaking complex process optimisation activities are core chemical engineering activities. Generating cost benefit analysis, assisting in precise manufacturing scheduling, responding to out of specification/process deviations and undertaking root cause analysis are all daily tasks of biochemical/biomanufacturing engineers.
Beyond batch manufacture
There is another avenue that is being explored, one which almost all other industries have purposefully marched down: continuous or intensified manufacturing. This concept involves a bioreactor operating with a perfusion operational methodology, constantly harvested, fed into a continuous purification train to separate and purify the delicate product. The process is controlled in real time by sophisticated AI-driven automation and digital tools. This is the end goal that the industry is striving towards, the continuous operating methodology that is ubiquitously employed by the manufacturers of everything from sugar to diesel. This coveted objective forms a core part of what is being referred to as Industry 4.0.
Achieving continuous manufacturing allows for significant reduction in COGs, capital, unit op size and increase in utilisation. However, there are some critical challenges which must be addressed to effectively implement this. Some key technologies are market ready with many pieces of the puzzle available or becoming available in the next couple of years.
The biopharmaceutical industry...will need talented chemical engineers from all backgrounds and industries to successfully implement manufacturing methods and philosophies widely used elsewhere
From the advances of understanding of biology and biochemistry to the design and implementation of modern production and purification systems, it is an exciting time for the biopharmaceutical industry. However, the industry will need talented chemical engineers from all backgrounds and industries to successfully implement manufacturing methods and philosophies widely used elsewhere. In the next issue we will review the state of continuous manufacturing in biopharma, what the challenges are and what new technologies are either in development or on the horizon.
References
1. www.historyofvaccines.org
2. Griffiths, E et al, “Polio vaccine: The first 50 years and beyond”, Biologicals 2006, 34, (2), 73-74.
3. Burrell, CJ et al, Chapter 11: Vaccines and Vaccination, in Fenner and White’s Medical Virology (Fifth Edition), Academic Press: London, 2017; pp 155-167.
4. Merck, Vaccine Bioprocessing Handbook, 2020.
5. Yongky, A et al “Process intensification in fed-batch production bioreactors using non-perfusion seed cultures”, mAbs 2019, 11, (8), 1502-1514.
6. Shukla, AA et al, “Evolving trends in mAb production processes”, Bioengineering & translational medicine 2017, 2, (1), 58-69.
©2022 Merck KGaA, Darmstadt, Germany and/or its affiliates. All Rights Reserved. Merck is trademark of Merck KGaA, Darmstadt, Germany or its affiliates. All other trademarks are the property of their respective owners. The Life Science business of Merck operates as MilliporeSigma in the US and Canada. Acknowledgement: Michael Burns, Paul Beckett, Stuart Rolfe, Carole Inglevert.
This is the second in an ongoing series of articles on how chemical engineers contribute to the biopharmaceuticals industry. To read the full series as it develops, visit: https://www.thechemicalengineer.com/tags/chemical-engineers-and-the-biopharmaceuticals-industry/
Recent Editions
Catch up on the latest news, views and jobs from The Chemical Engineer. Below are the four latest issues. View a wider selection of the archive from within the Magazine section of this site.




