Turning Points for IX

John Bewsey describes a new ion exchange process for cleaning up acid mines and brackish water
WE were delighted when the first ZIX-Zak (zigzag in ion exchange) treated water came from the final CSTR (continuous stirred tank reactor) of our demo plant in Krugersdorp, South Africa with a conductivity reading below 20 mS/m (equivalent to 30 mg/L sodium chloride) – better than the best grade of potable water, which has a limit of 70 mS/m (100 mg/l NaCl).
The US has made it illegal to dispose of reverse osmosis (RO) residual concentrate back into aquifers, and this ruling will spread world-wide as this practice destroys available water sources into the future. An answer for the use of RO inland is, thus, to build and maintain expensive lined dams to store this worthless saline effluent ad infinitum. At the coast the sea serves as a sink for this concentrate so RO will always be used for seawater desalination. However, a suitable solution for inland recovery of brackish and mines effluents is to use ion exchange, as it can separate the ionic pollutants into usable constituents and convert these extracted species into useful end products which ensure that there will be no liquid residual effluent problem.
Development
Trailblazer Technologies has spent two years building and commissioning our third-generation demonstration plant to convert acid mines water (AMD) and unusable brackish underground water (BUW) to usable irrigation and potable water, and product fertiliser and industrial salt. As these water sources are usually large and can feed continuous volumes – 15–120 ML/d – the use of column ion exchange technology was eliminated early on in our work. The pressure drop across columns are high so they require expensive pressure vessels and need large numbers of columns to treat the kind of flows required.
Ion exchange columns also use large amounts of fresh water to rinse and regenerate – of the order of 30 bed volumes (BV) per treatment, often making them net water users even at moderate dissolved solids content (TDS) and above. So we decided to operate using CSTRs, which allow for continuous, counter-current, low pressure operation and PLC controllability which is suited to large flow treatments.
As the average concentration of TDS in AMD and BUW varies between 3,000–15,000 mg/L we found that five stages of CSTRs for catex resin and three stages for anex resin processing was sufficient to achieve potable water recovery and profitable fertiliser manufacture. (Catex resins and anex resins attract cations and anions respectively.)
As our financial goal is to create fertilisers for resale from most of the regeneration chemicals, the logical raw materials to use for regeneration are nitric acid for the catex side and ammonia for the anex conversion. We used a strong-based cation resin and a weak-based anion resin to allow for the use of ammonia for regeneration. Strong-based anex resin requires sodium or potassium hydroxide to be used for regeneration and these raw materials are either too expensive or don’t convert to fertilisers to recover their value.
As our financial goal is to create fertilisers for resale from most of the regeneration chemicals, the logical raw materials to use for regeneration are nitric acid for the catex side and ammonia for the anex conversion
Due to the density difference of the ion exchange resins from the aqueous solution we are able to operate in a Pachuca style of overflow between CSTRs, removing filtration of the resin and some pumping between stages. Each tank stirrer is set to create a fluidised bed of resin in the CSTR up to around 80% of the reactor volume which gives a homogeneous fluidised bed of resin but allows a clean overflow for the aqueous phase. The resin from each stage is pumped to the forward CSTR using a peristaltic pump which controls the bed height and density via the main PLC controller. The water that overflows from one CSTR to the next is fed to the bottom of the following CSTR so that the aqueous phase always passes through the fluidised resin bed. 90% of full equilibrium is achieved in 7 minutes and this time factor is used to design the CSTR volume. The anex equilibrium time is much shorter, so fewer CSTRs can be used to achieve complete removal.
The regeneration of the spent resin is done in column fashion operated by an Ionex multiport valve which allows the regeneration to be achieved using only 1.2 bed volumes of regen chemicals and rinse water ensuring a concentrated product solution that keeps the cost of evaporation to a minimum. Column technology can be used here as the flow rate is usually around 6% of the feed water flow rate and efficient rinsing can be done using minimal volumes of water. The catex resin is regenerated using 20% nitric acid and the anex resin is regenerated using 8% ammonia. These regen solutions will consist of sodium, calcium and magnesium nitrate from the catex side and ammonium sulfate and chloride from the anex side. The regenerated and freshly-rinsed resin is pumped back to the last CSTR in the extraction bank on each side for the return deionising operation counter to the aqueous phase.
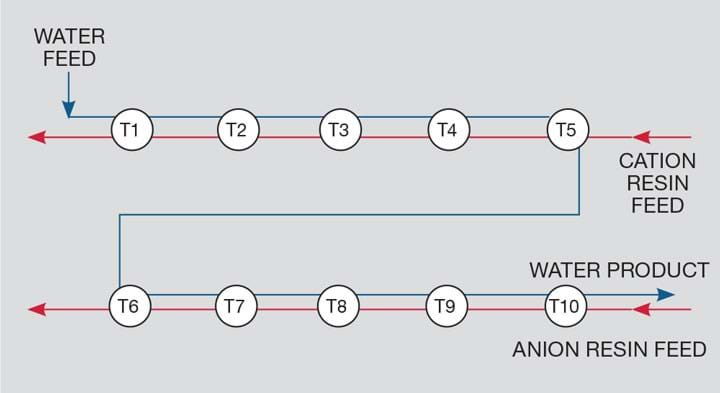
When the plant was operated in the initial, and more obvious, style of passing the aqueous phase first sequentially through the cation bank of CSTRs and, thereafter, sequentially through the anion bank of CSTRs we had the problem of a buildup of H+ in the final cation CSTR. Due to equilibrium between all the cation species this made it impossible to remove all the cations, and singly-charged cations like sodium remained in the solution. The weak-based anion bank couldn’t remove the anions associated with these residual cations so we were not able to reduce the TDS below 1,000 mg/L – being mostly sodium and chloride - which made the product water rather poor quality and not even fit for agricultural use.
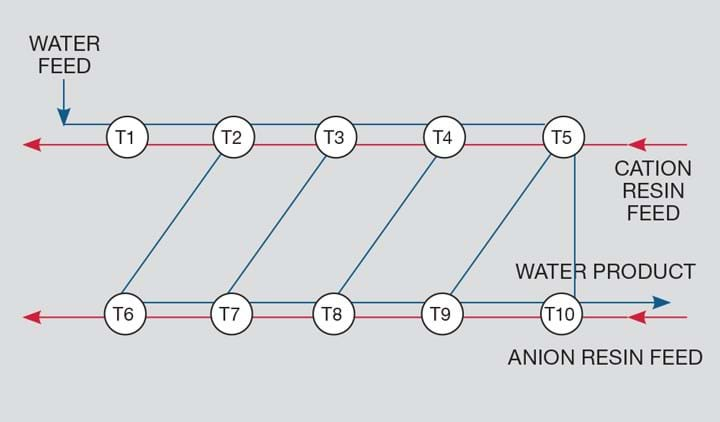
The zix-zak process
We then decided to run the aqueous phase in a zig-zag fashion between the cation CSTRs and the anion CSTRs which eliminated any buildup of free acid. We then produced water having close-to-zero TDS – effectively demineralised water. This water can then be chlorinated and fed directly into the main potable water system. This process has been patented under the name ZIX-Zak (Zigzag for ion exchange - IX) in several countries so far.
Recent Editions
Catch up on the latest news, views and jobs from The Chemical Engineer. Below are the four latest issues. View a wider selection of the archive from within the Magazine section of this site.




