The £300 Industrial Secret that Changed the World
Martin Pitt looks at how acquiring gunpowder know-how shaped the world as we know it
IN 1561, Queen Elizabeth I paid £300 (equal to about £100,000 today) to German engineer Gerard Honrick, for an industrial process “Instructions for making saltpetre to growe” – saltpetre being the primary ingredient in gunpowder. England had no significant natural source of this, so was short of gunpowder as she tussled with other European nations both locally and in the increasingly important colonies in the New World.
This began a whole series of historical changes with a huge impact worldwide today, with industrial chemical processes at the heart of them. It may well have been one of the most momentous actions in history.

Saltpetre – the “mother of gunpowder”
Saltpetre or saltpeter (also known as nitre) is crude potassium nitrate, the main ingredient of gunpowder (about 75%). The others are charcoal (available from English trees) and sulfur (mined in Iceland, so available). Natural processes led to deposits in various places, particularly hotter countries, and England traded as far as Persia, and employed smugglers to try to get more than other nations, in preparation for war. It has been compared to oil or uranium in strategic importance.
The secret was how to make saltpetre from pee and poo, (of which pee was the most important) in what was called a nitrary, by greatly accelerating the natural process and performing a crystallisation. Essentially, urine-soaked earth and animal manure was mixed with lime or potash and exposed to the atmosphere, turning at intervals. The mixture, depth of bed, and frequency of turning were part of the secret knowhow. Eventually crystals of KNO3 would appear on the surface in the same way that white minerals sometimes appear on the surface of damp brickwork, and would be scraped off, dissolved to separate out the earth, and recrystallised. (Because of their different solubility curves, KNO3 crystallises preferentially to NaNO3, though the latter also works in gunpowder, if the recipe is adjusted for the different mass.)
With this home-grown source, England was able to see off its European rivals. Elizabeth’s father, Henry VIII, had established a quality control system for armaments, and this was applied to gunpowder. With balls all being the same size and weight, gun barrels evenly and precisely sized, and consistent burning gunpowder, artillery and firearms were more accurate. This reliability under royal control was a major advantage for armed forces in Empire building.
Britain also developed “corning”, which we now call granulation, with wet grinding as used in the pharmaceutical industry today
Britain also developed “corning”, which we now call granulation, with wet grinding as used in the pharmaceutical industry today giving “black powder” where each grain (or corn) contained all the ingredients, giving even burning, and the corn size was suited for different weapons (larger for cannons). Particle technology was critical in the quality of the product in the Royal Gunpowder Mills. (Frederick Nathan, second President of IChemE, was Superintendent of the Royal Gunpowder and Small Arms Factories.)

The Saltpetre Men
The increased demand for saltpetre in the next century led King Charles I to order “his loving subjects carefully and constantly keep and preserve in some convenient vessels or receptacles fit for the purpose, all the urine of man during the whole year, and all the stale of beasts which they can save and gather together whilst their beasts are in their stables and stalls, and that they be careful to use the best means of gathering together and preserving the urine and stale, without mixture of water or other thing put therein. Which our commandment and royal pleasure, being easy to observe, and so necessary for the public service of us and our people, that if any person do be remiss thereof we shall esteem all such persons contemptuous and ill affected both to our person and estate, and are resolved to proceed to the punishment of that offender with what severity we may.”
There were daily collections in London in the Summer.
In addition, sources of urine-soaked soil and manure were gathered by the Saltpetre Men. In 1621 the country was divided up, and individuals were licensed to secure quantities from anywhere people or animals peed in their area. Barns, stables, dovecotes, sheep pens, the rear of alehouses, even the earth floors of theatres and village churches, where ladies took advantage of their long dresses and lack of underwear to find relief. Each main licence holder paid £1,700 a year, and employed teams to do the actual dirty work, with a weekly quota.
Initially this was supposed to be by mutual arrangement, but in 1646 the Saltpetre Men were given the power to dig anywhere, and owners who refused were prosecuted. Well-rotted areas were best.
In order to determine where to dig, and how deep, a Saltpetre Man tasted lots of soil and manure!

Why gunpowder goes bang!
This is the stoichiometric equation, but it works over a range of compositions.
8C + 3S + 10KNO3 = 2K2CO3 + 3K2SO4 + 6CO2 + 5N2
If you have hosted a barbecue, you will be aware that you cannot light charcoal with a match. Sulfur plays the part of a firelighter, being easier to ignite, and melts to contact the carbon better. The heat decomposes the KNO3 to give oxygen at a higher concentration than air, giving more intense combustion, and is not limited by diffusion of air through the bed of particles, as it would be with air as the oxidant. Energy is released, of course, but the key is that three solids give two solids plus eleven volumes of gas. This is what makes it a propellant in a gun. If it is contained in a vessel, then pressure energy is stored until the vessel breaks, giving a sudden release of that mechanical energy in a pressure wave – a bang and destructive effects. Heat energy adds to this effect .
Agricultural and industrial revolutions
As the illustration above shows, nitraries commonly had a problem with weeds. This led to an understanding of the importance of certain materials in the growth of crops. Nitraries produced material for explosives, but in 1672, just over 100 years after Elizabeth’s purchase, the East India Company began trade in Indian nitre, which eventually became 15% of its cargo, and one of its most profitable products. The alternating monsoons to leach material out of the soil followed by hot dry periods created natural crystallisers giving large deposits of soil with high KNO3 content which was carried to Britain for extraction and purification. The sewage smell of such a ship was well-known.
(The East India Company actually gained much of its trading advantage by force of arms with its own army and British firearms and ammunition. It eventually carried half of all world trade and effectively ruled much of India, until the British Crown took over in 1858.)
This trade meant there was enough to use as fertiliser as well, later supplemented by deposits in Chile and Peru, and nitraries were no longer used.
Crop rotation and use of this and other mineral fertilisers to provide N, P, and K compounds led to much greater agricultural productivity in Britain, meaning that many more people were available to work in industry. Machines were mainly powered by water. The Agricultural Revolution therefore gave the possibility of another major historical change.
By this time, much of the forests had been cut down for fuel and charcoal (for iron as well as gunpowder). However the discovery that coal (of which there was plenty) could both provide fuel and be converted into coke as an alternative to charcoal for iron meant that the Industrial Revolution took off in Britain, with the first blast furnace (a chemical reactor) in 1709. Coal-fuelled steam engines also provided power anywhere, unlike waterwheels.

Making Britain great
The head start in technology, and the established trade and military power made Britain the workshop of the world. One of the most important industries was textiles, where raw fibres could be imported and high quality cloth or garments exported at a cost far less than hand loom products. Ireland and Scotland mainly processed linen. In England the cotton trade was to the west of the Pennines, where higher humidity meant it was less likely to break. Manchester boomed, being nicknamed Cottonopolis. To the east was wool, with Bradford being the centre, and Leeds becoming a business centre with stock exchange, which it retained until 1973.
While history normally focuses on mechanisation, chemical treatment was the key to mass production.
While history normally focuses on mechanisation, chemical treatment was the key to mass production
Until the end of the 18th Century, bleaching cloth involved spreading it out in fields to be bleached by sunlight for up to 18 months. Various recipes involving stale urine, sour milk and lime or sulfuric acid were used to reduce the time to as little as four months, giving rise to a sulfuric acid plant for this purpose in 1746.
Scottish weaver Charles Tennant (1768–1838) and chemist Charles Mackintosh (1766–1843) – inventor of the waterproof raincoat – combined lime with chlorine to invent bleaching powder, calcium hypochlorite, which did the job cheaply and quickly, the first plant commencing production in 1801 at St Rollox in Glasgow. This expanded and was reckoned to be the largest chemical plant in the world in the 1830s.
Other plants of this design were constructed in Ireland (though they never paid any royalties) and England.
For other processes of cleaning and dyeing cloth, a chemical industry grew up particularly around the textile industry in the north of England, and this is where modern chemical engineering had its birth. Coal tar, being a byproduct of coke production, was the basis for the organic chemicals industry which followed soon after, especially the new coal tar dyes.

The Great War
In 1909 Germany was preparing for war. Nitrates for fertiliser and explosives were no longer made in nitraries, but imported from deposits of thousands of years of bird guano (poo) which had piled up in South America in addition to the Indian nitre soils. This resource had been a significant factor in colonisation of America. (The miners killed birds still trying to raise chicks there, and brought some close to extinction.)
Unfortunately for Germany, the main opponent in the war would be the British Empire, which controlled much of the nitrate sources and had the largest navy in the world to prevent access.

At last chemist Fritz Haber’s process for making ammonia out of thin air got the funding it needed, and chemical engineer Carl Bosch developed a heroic new sort of chemical engineering (huge high pressure, high temperature catalytic reactors). Their success also changed the world, though not immediately for the better.
While these new plants were being built, nitrates intended for fertiliser were used for explosives on a never-before-seen scale, causing crop yields to drop drastically, and the German people to go hungry. In addition, people from several nations killed each other in what was called the War to End All Wars – incorrectly as it turned out.
This First World War finally ended, and the Haber-Bosch process became the main supplier of nitrate fertiliser, reducing hunger around the world. As a bonus, some of the seabird populations were less molested and were able to recover their populations somewhat.
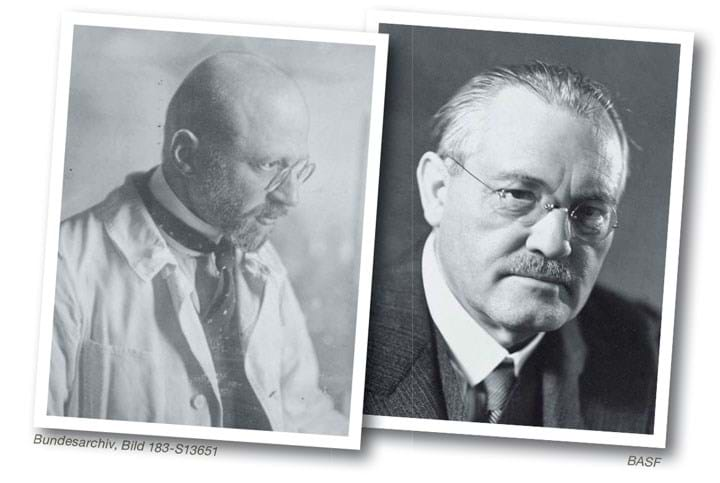
Conclusion
If Good Queen Bess had not bought this chemical secret then the British Empire would probably not have become the greatest in the world, and her colony of Virginia might not have expanded to create the USA. French might well have become the international language today rather than English, and quite possibly the language of England. The Agricultural and Industrial Revolutions would have happened elsewhere, and chemical engineering would have had another history. In short, the world would have been very different. In terms of value for money for the nation it was probably one of her best purchases.
For more on IChemE’s Centenary, including historical reflections from IChemE members, visit the dedicated website: www.chemengevolution.org
Recent Editions
Catch up on the latest news, views and jobs from The Chemical Engineer. Below are the four latest issues. View a wider selection of the archive from within the Magazine section of this site.




