Hydrogen: The Burning Question
Mike Menzies asks what effect does injected hydrogen have on furnace, flame and exhaust in natural gas combustion plant?
THE world is rising to the exciting challenge of controlling CO2 emissions. Replacing natural gas with hydrogen is progressing up the list of potential remedies for the domestic market.
The general public is increasingly aware of global warming, and remedies such as Reduce, Reuse, Recycle. But ‘Replace’ is being investigated with respect to the carbon in the National Grid gas supply. We had hydrogen in towns gas throughout the middle of the last century and the researchers are investigating replacing methane in the gas main with hydrogen, at least in part to begin with.
Hydrogen is well known in many large industries, but for production of other materials. The hydrogen is usually made from natural gas, by steam methane reforming (SMR) without capturing the CO₂, and in sufficient quantity for the process. Choosing one of the iron and nickel family of catalysts, together with heat, steam, methane and oxygen, a large proportion of hydrogen is made, together with CO2. Depending on production stability, the quantity of hydrogen may be in excess, in which case the plant looks to use the excess hydrogen elsewhere – usually by injecting it into the plant gas main. Hence this gas main composition changes from that of natural gas to include hydrogen, typically up to 30%.
What effect does the hydrogen composition have on the furnace, the flame, and the exhaust?
The good
The benefits of hydrogen include:
- it, like methane is not poisonous, (just asphyxiating and explosive);
- it has quite a high spontaneous ignition temperature (SIT) of 650oC – it needs a spark to ignite;
- it has very wide flammability limits (3–70% H2 in air mixture) – it is easier to maintain a flame;
- it burns to water vapour, thus eliminating CO2 emissions; and
- it burns with a much higher flame speed (300 cm/s) than methane (30 cm/s), thus stabilising the flame.
The bad
Disadvantages include:
- the higher flame speed increases the flame temperature locally, which can generate high levels of NOx;
- the wide flammability limits require consideration in the safety assessments;
- hydrogen has a different Wobbe Index from methane, to be taken into account in design (the Wobbe Index is a measure of the ability of a gas to deliver heat through a jet hole at constant conditions. It is calculated by the calorific value divided by the square root of the specific gravity of the gas); and
- hydrogen has a different combustion air requirement index, CARI (a measurement of the combustion air required for a gas), compared with methane.
Flames and NOx
This plant fuel gas is what the furnaces receive. So how does the hydrogen content affect the NOx emissions? The higher flame speed increases the flame temperature locally, which generates NOx. So the burner manufacturer has to design a burner to give a flame which will minimise the production of NOx. There are many ways of accommodating high hydrogen fuel gases whilst still keeping the flame cool enough to minimise NOx formation. The key is to slow down the rate at which the fuel and air mix. This gives rise to a diffusion flame. A diffusion flame is where the incoming gas is surrounded by the products of combustion where the gas and oxygen in the air have reacted. This sleeve or cloud of exhaust products slows the combustion because the gas has now got to diffuse out through the cloud, and the oxygen has to diffuse inward. Hence the name “diffusion flame”. This flame is usually slow enough such that the heat of combustion is radiated to the surroundings without reaching the critical NOx temperature of 1,350oC. The diffusion flame may well obtain sufficient temperature to crack the fuel before it is combusted. There will then be free carbon which will burn with a yellow flame. This does not look as well controlled as a sharp blue flame, so low-NOx burners, involving diffusion flames rather than deflagration, are only slowly being recognised by the operators. Many operators will remember bright yellow flames from heavy fuel oil or vacuum residue oil which had less acceptable exhaust composition.
There are many ways of accommodating high hydrogen fuel gases whilst still keeping the flame cool enough to minimise NOx formation. The key is to slow down the rate at which the fuel and air mix
The converse of a diffusion flame is a deflagration flame. In this case the gas and air are premixed and admitted into the combustion chamber, where they find a source of ignition. The flame propagates at the flame speed so the gases have to be injected at a velocity greater than the flame speed, otherwise the flame could “flash back” and burn in the burner mixing chamber. Premix burners therefore have a limited turn-down range and are designed for a particular flame speed. Adding hydrogen increases that flame speed and makes existing premix burners unsuitable for conversion to high hydrogen gases. The combustion rate can be reduced by diluting combustion air with exhaust gases as a method of slowing down the flame to reduce the temperature, but many installations have natural draft or induced draft premix burners where chamber negative pressure and gas aspirators entrain fresh ambient air into the premixer and thence to the burner quarl (prefired refractory burner block). So, we cannot reduce the oxygen partial pressure in these burners without modification to the air and associated control systems.

The flame which one hopes never to see in a furnace is a detonation flame. The heat of combustion from a flame at the centre of a pre-mixed cloud of gas and air produces a pressure wave which is sufficient to provide the source of ignition to the adjacent mixture (like a diesel engine) and so the combustion front proceeds at the speed of sound, or greater.
There is a further flame to consider - flameless combustion. This is used in high temperature furnaces where the burner design is not important. The gas and air are injected into the chamber separately and are heated by the radiation within the chamber to well above the SIT before mixing occurs. So wherever the gas and oxygen meet they will react and give off more heat. The exhaust gases will, of course, take heat out of the high temperature furnace and so heat recovery devices such as regenerators or recuperators become a necessity. The radiation from the flame appears less because the flame is dissipated throughout the combustion chamber where the light intensity is high – white hot. In these cases the furnace can be designed for the gases to wipe the crown of the furnace transferring heat by convection to refractory. The refractory has an emissivity near 1, so radiation to the product is achieved. Radiated heat energy incident on a surface is either absorbed or reflected. The amount that is absorbed is also re-radiated or emitted. The type and surface of all materials has a different proportion of reflection and absorption (emission). The two proportions always add up to 1 and the particular material can be simply described by the emissivity value which will be between 0 (shiny gold surface) and 1 (matt black surface). Regenerators or recuperators are used to recover heat from high temperature furnace exhaust gases.
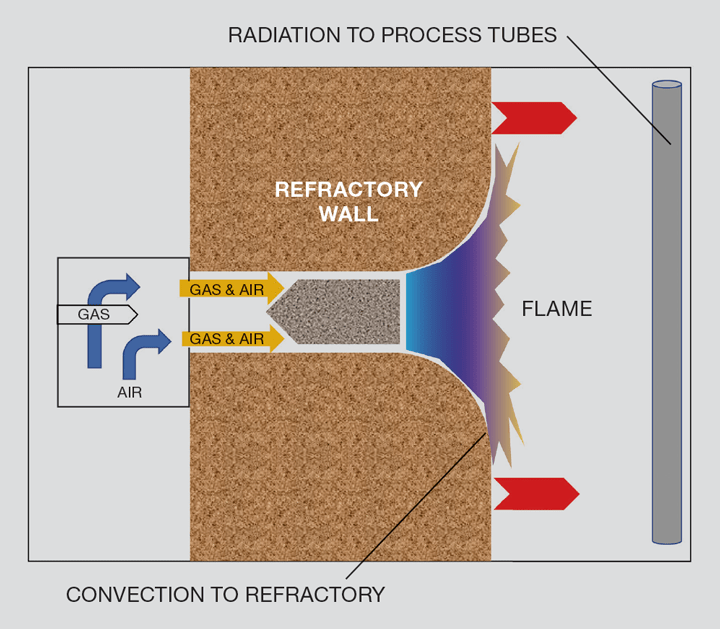
Natural draft premix burners
Returning to the natural draft premix burners, like the high temperature furnaces referred to above, they may well include design which promotes flame impingement onto the refractory to promote even radiative heat transfer from the heater wall to the product tubes. These small, wall-hugging burners use convection to transfer heat of the flame to the refractory using the Coanda effect or by nozzle design. (The Coanda effect results in a flow of gases, where these gases follow a curved surface because that curved surface has no gaseous molecules adhering to collide with – and therefore modify the direction of – the flow stream.)
Burner manufacturers have designs for both premix and nozzle mix flat flame burners, and for both induced draft and forced draft.
They may be suitable both for high hydrogen as well as low NOx. Adapting these burners for low NOx will often involve separating the flame into a rich and lean section where both parts of the flame have a lower flame temperature because of being away from the stoichiometric region. The rich zone may be as simple as a neat gas poker firing onto the edge of the lean gas flame. High hydrogen is ideal for these burners because of the wide flammable range of hydrogen in the lean zone and the high flame speed for stability of the rich jet.
The processes they are used on tend only to require a very limited turn-down range which allows a significant proportion of hydrogen in the fuel without incurring flashback on a premix burner. There are usually many burners, often several hundred, arranged in rows. During heatup after a shutdown, rather than trying to modulate the burners, gentle heat input is often achieved by lighting rows or individual burners on/off. Nozzle mix flat flame burners should not have any significant problem as the hydrogen content of the gas is increased, save for the choice of nozzle material. But the problem for premix flat flame burners is more significant with high hydrogen. A flashback might extinguish but will probably reoccur and if the flame stabilises at the premixer (a burnback) then the burner is likely to be destroyed in a short time, almost certainly before it can be observed by an operator. Fitting flame detection on all, say 500 burners, is likely to be blocked by financial considerations.
Process stability would suggest that modulation, even if it is only 2:1, would be beneficial. However, the percentage of hydrogen would be the limit to the range of modulation. Modulation of 2, 3, or 4:1 may be suitable with a methane gas but not for pure hydrogen. Phasing over from pure methane to high hydrogen could be achieved with new jets/nozzles, but this procedure would be suited to a specific change of fuel and not just to gradual changes in fuel composition.
Unlike the flat flame wall burners above, many boiler/heater/furnace/kiln low-NOx burner designs separate the combustion air into two parts – the primary air and the secondary air. The gas will be delivered in the centre with the primary air, and pilot flame or igniter (and often flame detection equipment). The secondary air is delivered outside the diffusion flame package, with low turbulence to minimise mixing. Power station burners tend also to be suited to low NOx and high hydrogen. In these power station burners there can be several other fuels. It’s not unknown for a boiler burner to have five different fuels, though one or two of these fuels may merely be a method of disposing waste or vent gas from another process. Coke oven and blast furnace gases are common, but they do have a significant influence on the original design of the burner. It is likely that one of the fuels may be “plant gas” shared by energy consumers throughout the plant.
These boiler burners are generally well suited to utilising high hydrogen gases in a nozzle mix arrangement. Furthermore, the control system fitted on a boiler is often sufficiently well instrumented that air:fuel ratios can be continuously controlled allowing gas composition changes without notice. To achieve low NOx, one of the favourite procedures is to reduce the combustion air to all burners (making the flames sub-stoichiometric) and removing all the fuel lance internals from the last row of burners, thus supplying the “secondary” air at the end of the combustion chamber. This design will need a further chamber for the final combustion (probably flameless combustion) before the exhaust gases pass into the convection section.
Calculation or measurement of the Wobbe index can be undertaken and is both a simple and effective way of trouble-free inclusion of higher levels of hydrogen gases. The Wobbe Index is a measurement of the ability of a gas to deliver heat through a certain size jet, but this may require a different amount of air to achieve it. So even if you can get the same heat out of a burner, you may need to put in more combustion air. This calculation is very similar. The combustion air requirement index, CARI, is the amount of air required to combust the gas so that the air stays in ratio with the gas. It is common for such a gas to be continuously monitored in a Wobbe index analyser.
There are many other matters to consider. For instance, when we put high levels of hydrogen in the fuel, the exhaust could turn white with condensing steam droplets as it cools. So, the public may see a change in the stack plume.

Summary
This review has looked at the development of burner design to take account of emission regulations, specifically NOx. In the main, this requires separating the combustion air into primary and secondary streams so as to achieve a diffusion flame. This diffusion flame is larger, slower, and cooler than its predecessor, the deflagration flame. So, because changing to low NOx will probably require new burners, it would make sound economic sense to choose a burner design which is also suitable for high hydrogen.
Your plant is likely to be legally obliged to control the NOx emissions. But in your neighbourhood and your nation you should be morally obliged to prepare for hydrogen
Your plant is likely to be legally obliged to control the NOx emissions. But in your neighbourhood and your nation you should be morally obliged to prepare for hydrogen. It’s not difficult. Burners can be designed for any gas composition with hydrogen. It is just a little bit more difficult if the composition changes excessively.
But undoubtedly, hydrogen is our friend in the furnace. We just have to control it.
This is the 11th article in a series discussing the challenges and opportunities of the hydrogen economy, developed in partnership with IChemE’s Clean Energy Special Interest Group. To read more from the series online, visit the series hub.
Correction: Figure 3 has been changed as it originally made an incorrect reference to "secondary air plus 100% gas".
Recent Editions
Catch up on the latest news, views and jobs from The Chemical Engineer. Below are the four latest issues. View a wider selection of the archive from within the Magazine section of this site.






