Honey, I Shrunk the Antimicrobial Resistance

IMAGINE needing lifesaving surgery. Let’s say an organ transplantation or to put you back together after an accident. Now think how you would feel if you were told: “sorry that intervention is just too risky, you may pick up a deadly superbug in recovery”.
That would probably lead you to ask why. I mean, we have all these fantastic healthcare technologies, surgical robots, metallic implants, pacemakers, chemotherapeutics, surely there are options to save your life? The truth is, in the post-antibiotic apocalypse many medical interventions, even relatively common ones (hip replacements, cancer treatment, caesarean sections), will become incredibly risky or technologies of the past. England’s chief medical officer, Dame Sally Davies, has put it quite simply: if antibiotics lose their effectiveness it would spell “the end of modern medicine”.
As a trained engineer I have no intentions of taking up a career in discovering and developing new antibiotics – there are definitely plenty of scientists out there who are better equipped for that challenge. In fact, the University of Birmingham has a history of research excellence in antimicrobial resistance (AMR), which includes experts such as Laura Piddock, director of Antibiotic Action and a leading voice in the debate about the lack of new antibacterial treatments. As engineers, we are often lured in by the challenges involved in overcoming societal problems and this is what motivated me to think about how our team could contribute. This is how I became involved in research to come up with effective delivery methods for a new honey-inspired antimicrobial, bioengineered by Matoke Holdings.
The project has attracted a lot of interest, and is the focus of fundraising activities this year, so read on to find out how we got here, and how you could help fight the war on superbugs.
What is antimicrobial resistance?
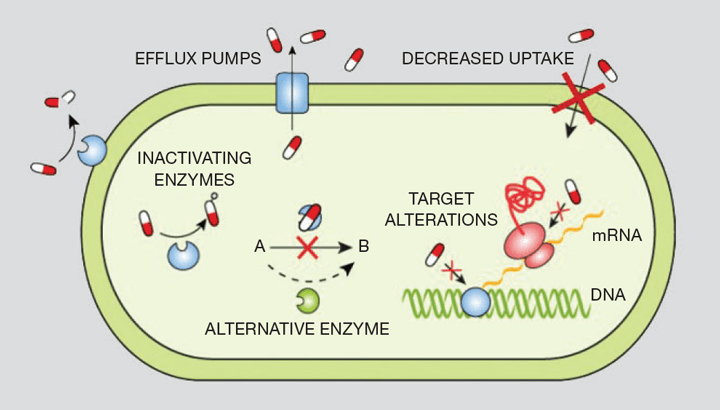
Ever since Alexander Fleming discovered penicillin in 1928, antibiotics have revolutionised the treatment of many previously deadly diseases, such as pneumonia and tuberculosis. Today, antibiotics are a cornerstone for preventing and controlling the development of infections in many parts of our bodies. However, throughout history bacteria and other pathogens have fought back by continuously evolving mechanisms to withstand the actions of antibiotics and other antimicrobial drugs. One of the main ways bacteria do this is by stopping the antibiotic reaching its target at a high enough concentration. Bacteria can achieve this, for example, by transporting the drug out of the cell through channels called efflux pumps that sit in their membrane or cell wall (see Figure 1). A colleague, Jessica Blair, once described this as “the leaky bucket problem” – while we are busy pumping more antibiotic in, the bugs are hard at work pumping it all back out.
The other major bacterial defence mechanism is to modify the target on which the antibiotic acts. In fact, this is how the well-known superbug MRSA (methicillin-resistant Staphylococcus aureus) works. Through expressing a new penicillin-binding protein with low affinity to b-lactam antibiotics, this prevents targeting of the bacterial cell wall.
How big is this problem?
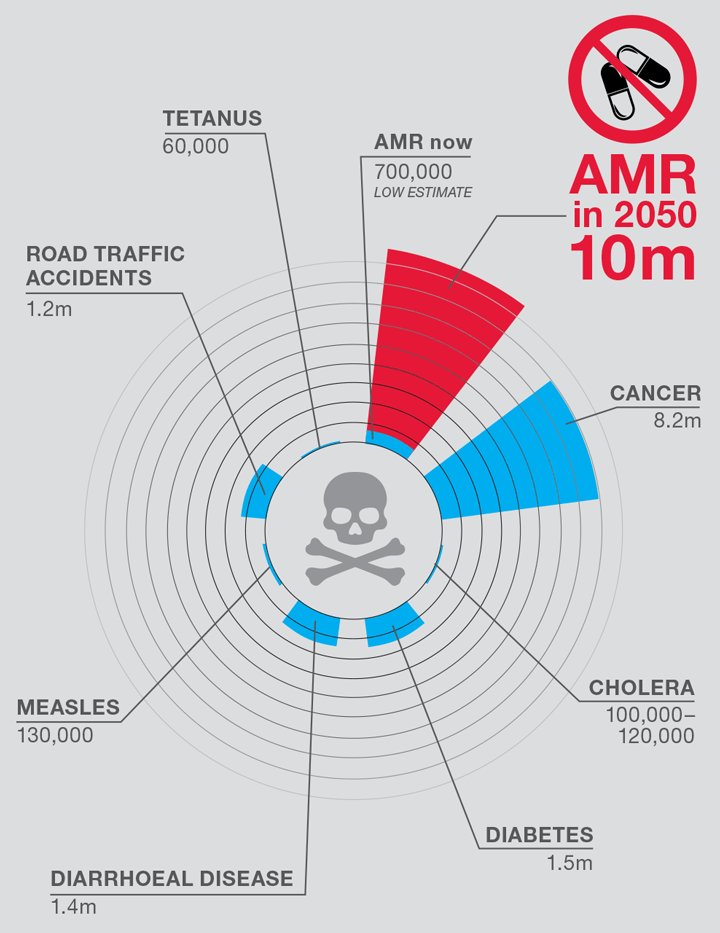
Today, an estimated 700,000 deaths per year worldwide are attributable to AMR. Compared with cancer (8.2m), diabetes (1.5m), and road traffic accidents (1.2m) these numbers are relatively small (see Figure 2), which has somewhat overshadowed the looming threat of AMR. However, in 2014, a review1 led by economist Lord O’Neill concluded that if we continue on our current trajectory, a continued rise in resistance would lead to 10m deaths per year from resistant microbes by 2050, which would cost the world economy an estimated US$100trn.
Recent Editions
Catch up on the latest news, views and jobs from The Chemical Engineer. Below are the four latest issues. View a wider selection of the archive from within the Magazine section of this site.




