Molten metal for clean hydrogen
MOLTEN metal could be used for a cheaper, more environmentally-friendly alternative to produce H2 from CH4, according to researchers.
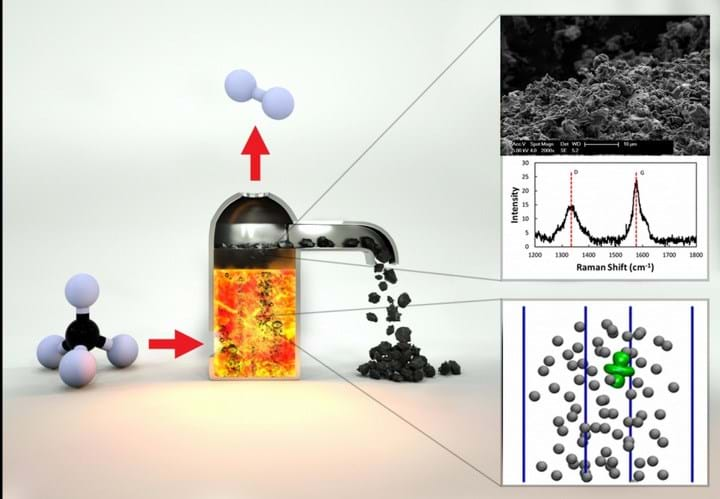
Currently, hydrogen used to make ammonia and other industrial reactions is produced mainly through steam methane reforming, using nickel catalysts. This is a high-temperature process, which releases CO2, often to the atmosphere.
But a new approach, developed by US and Indian researchers, instead produces a solid form of carbon which can be easily transported and stored indefinitely.
It does so through a single-step method, which uses nickel dissolved in molten bismuth. This pyrolyses CH4 to release H2 and form CO2, which floats to the surface of the melt where it can be skimmed off.
“You introduce a bubble of methane gas into the bottom of a reactor filled with this catalytically active molten metal,” said University of California, Santa Barbara chemical engineering professor Eric McFarland, adding: “As the bubble rises, the methane molecules hit the wall of the bubble and they react to form carbon and hydrogen.”
In a Science paper, the team of researchers demonstrated that a 27% Ni–73% Bi alloy achieved 95% CH4 conversion at 1,065°C in a 1.1 m high bubble column, producing pure H2. It also found that the molten metal alloy surfaces were not deactivated by accumulation of carbon, and could be reused indefinitely.
Using an active liquid metal highly soluble to H2 could also allow the process to be efficient with very high-pressure CH4, to produce high-pressure H2. McFarland said: “You’re really allowing yourself to pull all the products away from the reactants and that causes the equilibrium to be shifted toward the products. The process in principle can operate at high pressure and still get very high methane conversion.”
McFarland is confident that the team’s technology can be readily deployed using existing infrastructure, and said that it has captured the attention and support of Royal Dutch Shell.
Science: http://doi.org/cgkh
Recent Editions
Catch up on the latest news, views and jobs from The Chemical Engineer. Below are the four latest issues. View a wider selection of the archive from within the Magazine section of this site.




