Metal-free catalyst for ester synthesis
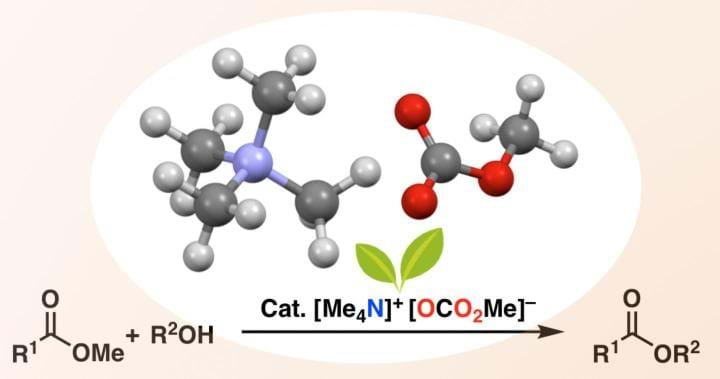
A METAL-FREE and recyclable catalyst has been developed for synthesising complex esters which could be used for biodiesel production.
Complex esters have a wide variety of industrial uses, ranging from moisturisers to biodiesel, and are produced by reacting simpler esters with alcohols in a process known as trans-esterification. This process requires a metal salt catalyst which can be polluting and/or expensive. The catalyst can also have the opposite effect and shut down the reaction. This can happen if the ester is long and flexible and wraps around the metal centre, in a process known as chelation.
Researchers at Nagoya University, Japan, have developed a metal-free catalyst that avoids chelation and therefore expands the range of esters and alcohols that can be used for trans-esterification. The catalyst is called tetramethylammonium methyl carbonate (TMC), and it reacts with an alcohol to form an alkoxide ion. The alkoxide ion then attacks the starting ester to create the complex ester. While this is not the first process to use alkoxides for trans-esterification, it is more versatile.
"Previous efforts have used phosphonium salts, but the resulting alkoxide can only esterify the solvent itself, which must be dimethyl carbonate (DMC)," said Manabu Hatano, co-author of the study. "That's fine if you want a product that can be derived from DMC, but otherwise you need salts that are stable in more conventional solvents. Our new TMC fills that gap."
"Being both recyclable and free of metals, this catalyst is a genuine example of green chemistry," said Kazuaki Ishihara, lead author of the study. "Not only is the process itself green, but we can use it to produce green biodiesel, which is a mixture of esters. We synthesised hundreds of grammes of a major biodiesel component, which is quite copious for a laboratory-scale reaction. This gives us confidence that the reaction can be scaled up to industrial production."
Green Chemistry http://doi.org/cmbm
Recent Editions
Catch up on the latest news, views and jobs from The Chemical Engineer. Below are the four latest issues. View a wider selection of the archive from within the Magazine section of this site.




