The Challenges of Developing a Fusion Fuel Cycle: and How Chemical Engineers are Helping to Make Fusion Energy Sustainable
In the final part of our series on fusion energy, Mirjana Damjanovic and Lewis Simmons highlight the significant challenge of decommissioning and repurposing a fusion powerplant
Once a fusion plasma is seen, it is not easily forgotten. That unique iridescent glow which fills a tokamak is a clear sign that powerful and impactful science is taking place, and an inspiration for aspiring fusion engineers to contribute towards the development of a potentially crucial piece of the jigsaw to achieve net zero.
The Joint European Torus (JET) at the home of the UK Atomic Energy Authority (UKAEA) in Culham, Oxfordshire, has staged well over 100,000 plasma pulses during its 40 years of operation, each of which has contributed a wealth of data to advance the understanding of plasma science.
At the end of 2023, JET’s plasma science operations will cease, and the transition will begin into a world-first decommissioning and repurposing programme which will last until around 2040 and be just as significant as the decades of plasma science that have preceded it.
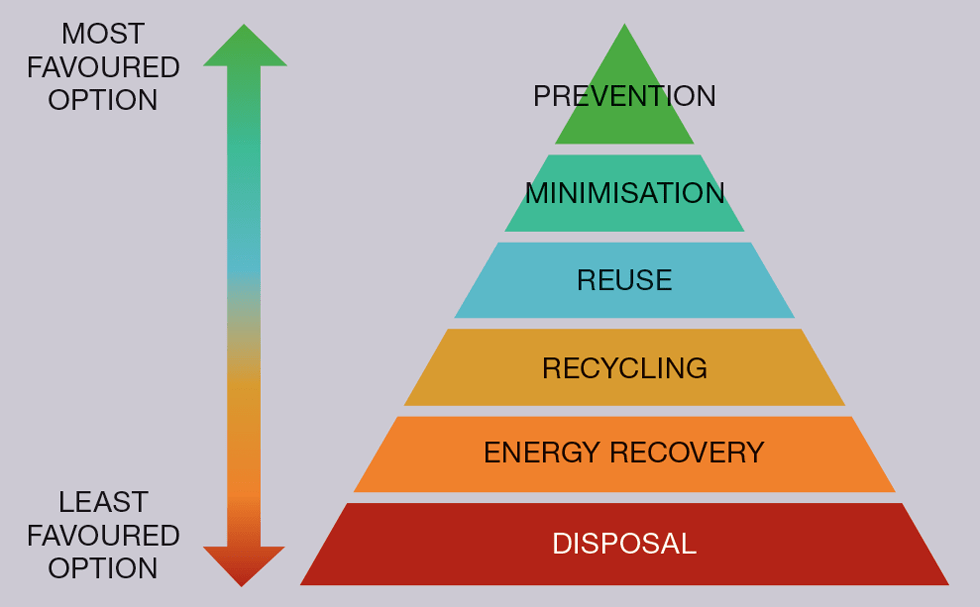
It is often said that fusion is a clean energy, but there is a missing end to that sentence: “because we apply the appropriate waste treatments and processes”. JET’s decommissioning and repurposing (JDR) is built around the principles of the waste hierarchy, whereby prevention, minimisation, re-use, and recycling is all prioritised within the design and operation of a future machine over disposal, the last option to be considered.
JDR is a significant challenge, both for the programme team and UKAEA as a whole. No fusion machine has been repurposed and decommissioned in the way UKAEA is planning.
It represents the missing piece for fusion. We need to prove the validity of our repurposing and decommissioning process to help make fusion as a whole deemed effective. The entire fusion community will be following everything we do and, while that is daunting, it is also exciting.
Fusion at JET
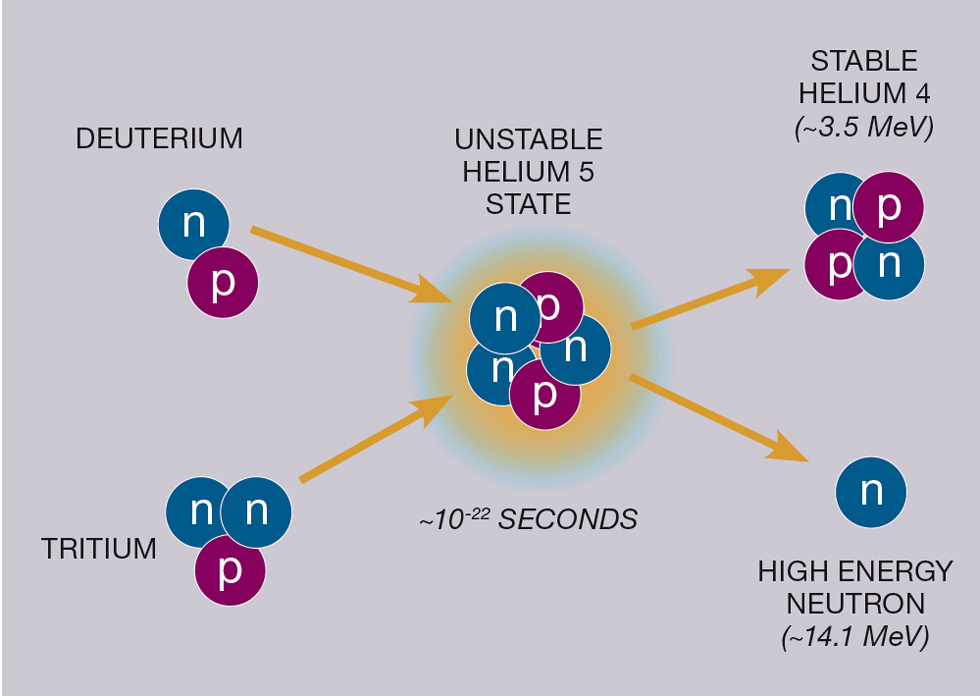
Most current pursuits for fusion power are reliant on fusion of two isotopes of hydrogen known as deuterium (D) and tritium (T). Deuterium is abundant, but tritium is rare and will need chemical engineers to help manufacture it at scale (see TCE 988). This is what has made JET’s research important, as it has been the only tokamak to use this mix of fuels.
They are chosen for fusion because the reaction is energetically favourable (easiest to achieve) compared with alternatives (and there are many different reactions available!). This ultimately means the temperatures required for D-T fusion are a lot lower than other fusion prospects, although still at a balmy ~150m °C.
Despite the D-T fusion reaction being the most technologically feasible, there are key implications for any materials which are near to fusion reactions. And while JET’s pulses last for a fraction of the 24-hour, seven-day operations of a power plant, there are nevertheless some key lessons for how materials behave under fusion conditions before large-scale facilities are designed and built.

The waste challenge
Tritium is a radioactive form of hydrogen with a half-life of approximately 12.3 years. Like all hydrogen isotopes, tritium can migrate into the atomic structure of materials via simple diffusion mechanisms which, combined with decay, causes these materials to become radioactive. At elevated temperatures, this diffusion through materials is significantly more effective, a key consideration given the very high temperatures within fusion machines. In summary, tritium has a tendency to spread wherever it wants and get stuck inside materials.
The maintenance and decommissioning of fusion machines will generate metallic waste containing high levels of tritium, classified as intermediate level waste (ILW). In addition to hard, tritiated ILW, operational, maintenance, and decommissioning stages of future fusion machines will generate high volumes of tritiated housekeeping waste such as gloves, protective suits, plastic sheeting, waste bags, etc.
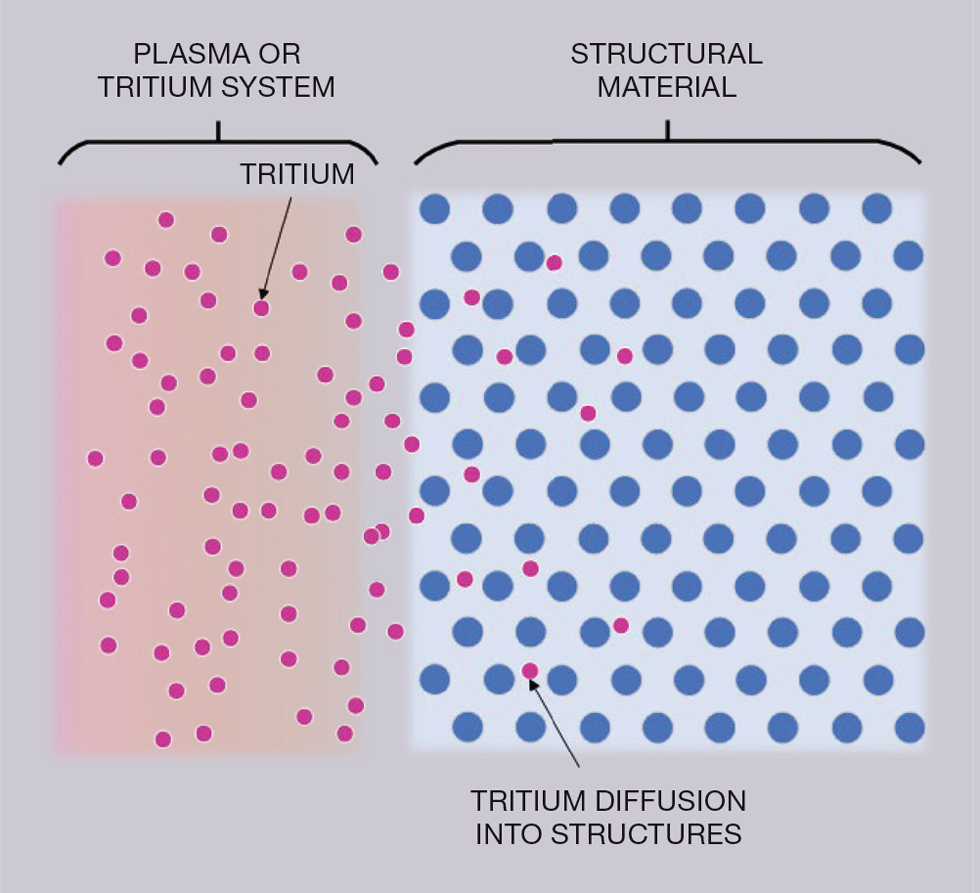
The nature of tritium makes it an interesting technical challenge. It is a radioactive gas, volatile, and able to penetrate and escape materials at different rates, and because tritium emits low-energy beta radiation it is difficult to measure. Furthermore, to assess tritium in a solid sample, we need to destroy that sample. This prevents two truly identical measurements, leading to a level of assumption.
Storage of tritiated waste poses environmental, health, and safety challenges, as tritium is constantly outgassing from the materials. Decay storage of tritiated ILW also means leaving a burden for future generations, something which fusion promises to minimise. Moreover, the valuable materials used in fusion could be recycled for future use.
The nature of tritium makes it an interesting technical challenge. It is a radioactive gas, volatile, and able to penetrate and escape materials at different rates, and because tritium emits low-energy beta radiation it is difficult to measure
Activation
The high-energy neutrons produced from D-T fusion are not charged particles and hence are not kept confined by the magnetic fields inside a fusion system. Instead they are free to travel out of the plasma and impact the surrounding structural materials. This is ideal for a power plant since plasma energy is transferred via neutrons, and heat can be captured.
However, the first thing that neutrons usually interact with is whatever the reactor is made from, usually a metallic material. When a neutron interacts with a nucleus in a material, it can be absorbed and change the state of the nucleus. This frequently results in an unstable nucleus being formed; this then decays, releasing radiation.
The more materials are exposed to neutrons, the more unstable nuclei are formed, increasing radioactivity. Furthermore, since metallic structural materials such as steel and other superalloys have many constituent elements, activation behaviour is incredibly complex.
Radioactivity and radiation
The tritium permeation and the activation of materials by fusion neutrons leads to the formation of radioactive materials in fusion system.
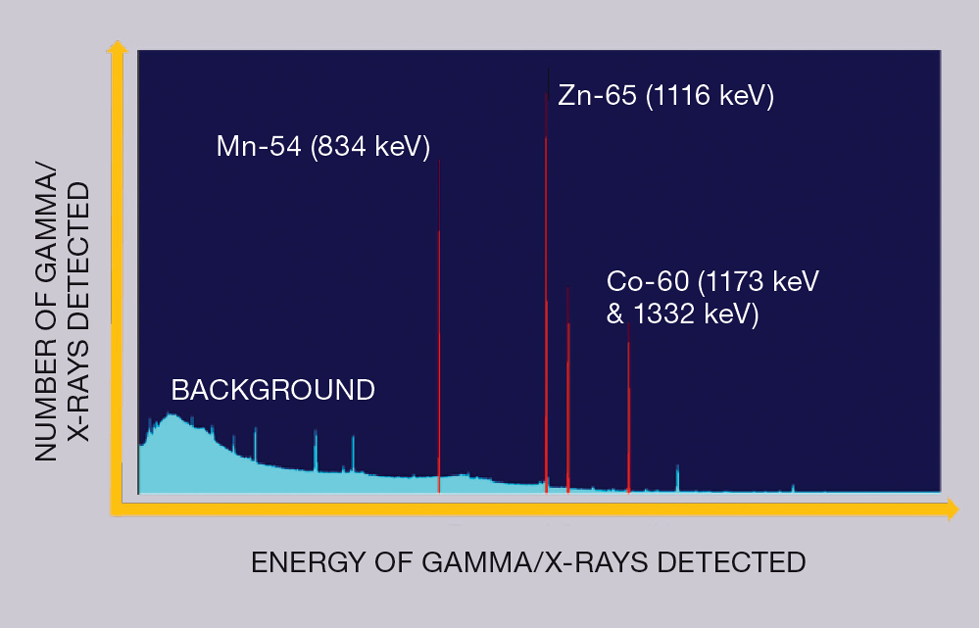
We frequently refer to alpha (), beta (), and gamma () decays in nuclear physics. Fortunately, almost all fusion-related radioactive materials decay via beta decay and then sometimes a subsequent gamma decay to release any excess energy. Cobalt 60 decay is a good example of this (see Figure 4) – it’s one of our key radioactive elements (radionuclides) from our JET steels.
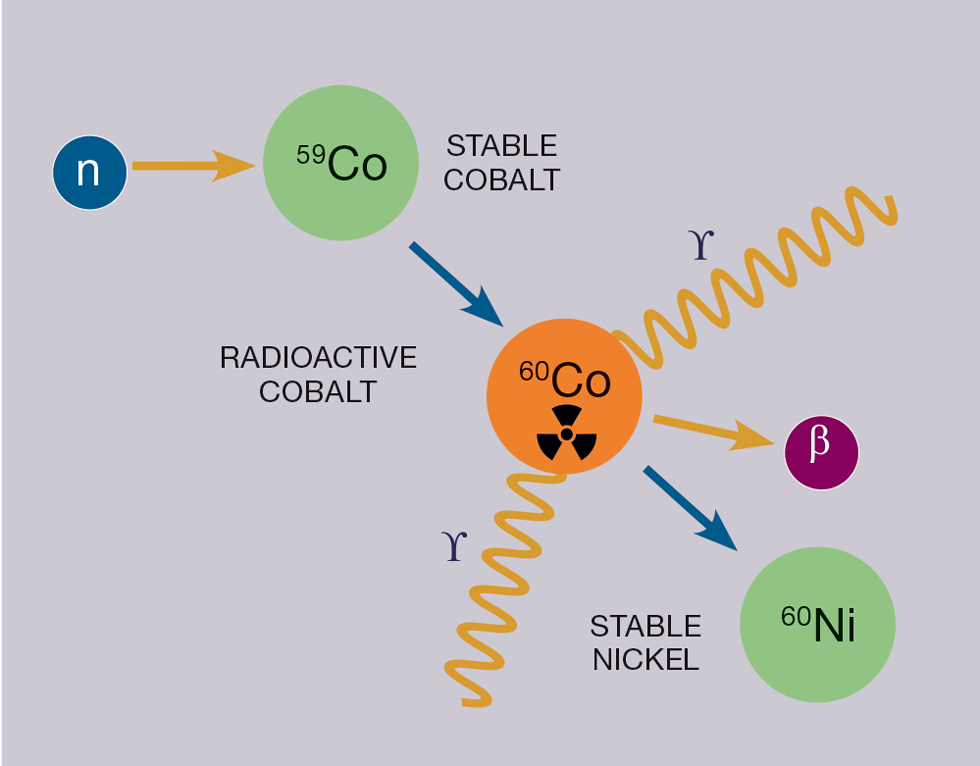
Each different radionuclide has a unique emission signature in terms of radiation, so it is possible to get an idea of which radionuclides are present in a material via specialised analysis – we refer to this process of identification as “characterisation”.
For example, several different metals – iron, cobalt, nickel, etc – are used in common structural materials like steels. JET is made of thousands of tonnes of this material and it gets bombarded with neutrons when we run plasmas. We get a whole host of different radionuclides forming which we need to get measurements of to dispose of radioactive waste effectively:
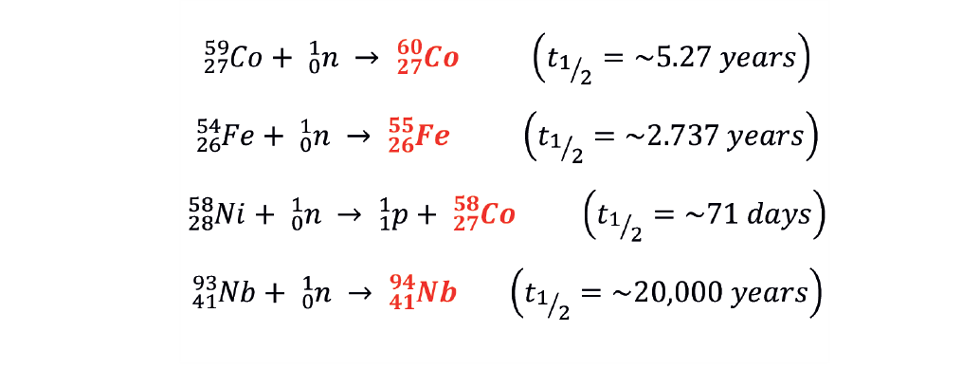
Tritium also migrates into materials such as steels, and decays according to its half-life:

Each of these radionuclides decays in a specific manner via the emission of particles of a certain type with a specific energy. The decay behaviour of thousands of radionuclides has been measured to create large libraries of decay information for us to reference. By carefully measuring the decay radiation and the associated energy released from radioactive materials, we can determine which radionuclides are present and how much of them we have in a material. This is vitally important since radioactive wastes are classified by the amount of specific radionuclides present.
Process for radiological characterisation
Gamma rays and X-rays
Since gamma rays and X-rays easily penetrate most materials and freely travel through air, placing a measuring device near to radioactive materials enables both to be measured. We place our materials in the vicinity of a gamma spectrometer, which measures which gamma or X-rays are produced, and their energies. Since each radionuclide emits X-rays or gamma rays with a specific energy, it is possible to identify which radionuclides are present. In addition, by assessing the relative numbers of different energy gamma or X-rays found, we can also determine the amount of each radionuclide present.

Alpha and beta particle emissions
Alpha and beta particles behave differently to gamma rays, so we can’t simply place our materials near a specialised detector to assess for beta and alpha activity. Instead, we must take samples of materials and use lab-based assessment.
After cutting, cropping, or sawing a small sub-sample from our radioactive material – no easy task – we usually have to get it into a fluid. Many of the common ways of separating out radioisotopes rely on chemical separation – exactly like chemistry classes at school. This is a huge-scale endeavour in terms of characterisation because we have so much material due to JET’s size. Once we have separated out radionuclides, we present them to specific detectors which hunt for alpha or beta particles, or to devices which assess radionuclide masses.
Though tritium releases beta particles, these are of sufficiently low energy that they simply cannot penetrate into a detector material and be measured. Therefore, the most common analytical measurement used in characterisation for tritiated materials is pyrolysis. Small samples of a material are rapidly heated in a tube furnace to thermally degrade them and release any tritium. This gaseous release is then passed into a capturing fluid (usually water) and analysed via a special liquid-based process known as liquid scintillation counting.
Combination of information
By deploying sufficient predictive modelling, selecting relevant radionuclides of interest, assessing which analytical techniques are needed, and then performing analysis, we can generate a radionuclide description for materials.
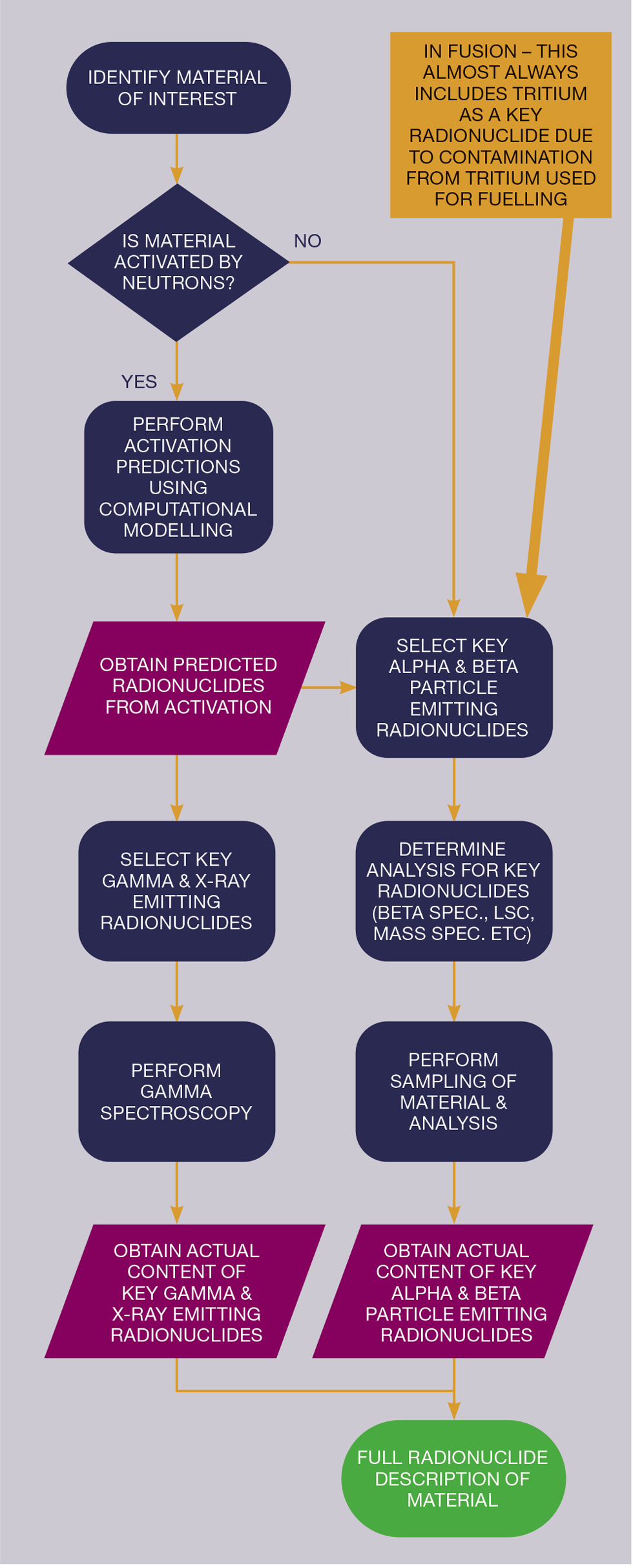
Since each radionuclide emits X-rays or gamma rays with a specific energy, it is possible to identify which radionuclides are present...we can also determine the amount of each radionuclide present
Radioactivity and radioactive waste
There are strict regulations regarding the safe storage, treatment and disposal of radioactive materials and regulatory bodies (such as the Environment Agency) enforce correct practices. In the UK, materials are classified according to how much radioactive material they have per mass of waste. It is therefore crucial to characterise materials, not only to understand their hazardous properties and keep people safe, but also to determine how we manage and dispose of our wastes.
Fortunately, the fusion process produces very few long-lived radioactive wastes, and does not produce high-level waste (HLW). With JET’s decommissioning, there is the opportunity to manage wastes in a manner which enhances the sustainability of fusion compared with other nuclear applications. These include:
- decay storage of wastes, such that radionuclides naturally decrease until materials can be reused
- the recycling of fusion materials into other nuclear applications (like future reactors) because of their relatively low radiological hazard
- the innovative treatment of wastes to remove tritium contamination (detritiation) and the down-categorisation of materials from ILW to lower waste categories
Detritiation
As already discussed, tritium contaminates systems associated with plasmas and fuelling. The level of radioactivity within these systems would make them ILW once they are decommissioned, and there would need to be a body responsible for the maintenance of ILW for more than 120 years.
Detritiation, which is the process of removing tritium from materials, offers multiple benefits:
- environmental benefits by reducing the tritium off-gassing from the materials
- safety by reducing the exposure of personnel handling ILW
- reduction in the cost of disposal by reducing the total activity of waste below 12 kBq/g. This would open a low-level waste (LLW) disposal route for hard waste
- detritiation of housekeeping waste will reduce incineration costs considerably
- characterisation benefits for soft waste by detritiation as information builds about the efficiency of detritiation techniques and tritium recovered fuel recovery and reuse/recycling of the materials. Capturing the tritium removed from the waste will improve tritium accountancy and close the fuel cycle
- materials from which tritium was removed will have better potential of being reused and recycled following the waste management process
Within JDR, a research and development project is ongoing, with the aim of developing a “detritiation cookbook” for materials expected to be classified as ILW following dismantling activities.
UKAEA has demonstrated that detritiation of some JET materials may be highly effective using thermal methods. Therefore, the detritiation recipes are mainly focused on exploring heating conditions under which materials can be detritiated, preserving as much as possible of their structural integrity, ensuring safety of staff, and reducing environmental impact.
Moreover, the project is looking at the potential of processing different materials together and other aspects which would potentially allow increased throughput of the detritiation facility.
All the findings from the experiments and suggested recipes will be summarised in a detritiation cookbook, which will underpin waste processing and the design of future detritiation facilities, as well as the strategy of waste operations during the decommissioning period of JET.
The success of the project will not only enable high throughput and efficiency for JET waste processing, but also provide know-how for future fusion machines, which will produce significantly larger volumes of ILW during operational, maintenance and decommissioning periods.
Future impact
The direction which is being taken by UKAEA recognises the unique opportunities presented by the end of plasma science operations at JET.
For chemical engineers, opportunities to have an influence not just on a particular project or programme, but also the future direction of a global sector which has the potential to significantly impact the road to net zero, are extremely rare. Everyone within JDR understands the importance of the work we are undertaking.
The success of JDR requires critical thinkers who can assess and develop processes for materials removed from JET and its systems. The core skills developed by chemical engineers are highly applicable to waste and characterisation work and complement a multi-disciplinary team which includes engineers, mathematicians, and materials researchers. Over the next 18 months we will be tested by work beyond anything we have done previously in terms of radiological hazard and comprehensive sampling, scientific analysis, and ultimately, characterisation.
In considering the options for decommissioning JET, UKAEA has taken the decision to maximise the scientific and economic benefits, which will provide the designers and developers of future fusion power plants with insight and information crucial to their important task, and in doing so help to bring sustainable fusion power one step closer.
Engineers solving fusion challenges: Mirjana Damjanovic

My background is in engineering, and after completing a bachelor’s and master’s degree in biomedical and environmental engineering, I pursued a master’s in nuclear decommissioning and waste management. I really fell in love with nuclear engineering in general. I realised the benefits that nuclear energy can offer, and the challenge related to radioactive waste management and decommissioning was something I wanted to tackle. After I completed a master’s degree, I was working at a company that was collaborating with UKAEA, and under that collaboration I had the opportunity for a placement here tackling challenges related to radioactive waste from fusion. I’m really excited to now be a part of the first-of-a-kind decommissioning programme and be able to apply the knowledge and skills I gained.
Recent Editions
Catch up on the latest news, views and jobs from The Chemical Engineer. Below are the four latest issues. View a wider selection of the archive from within the Magazine section of this site.




