Hot Topic
Keith Plumb gives an overview of batch heat transfer
THE purpose of this article is not to provide methods to allow a detailed batch heat transfer calculation to be carried out, because this is not possible in a single article and could take a series of articles, the chapter of book or conceivably the whole of a book.
Instead I provide an overview of the factors that need to be taken into account and to allow the reader to have better understanding of their particular problem so that they can set off in the correct direction to carry out the required design.
Fundamentals of batch processing
Apart from the obvious point that batch processing involves the manufacturing of a discreet quantity of material (the batch), the fundamental point for chemical engineering design is lack of steady state and that the parameters for design are changing with time.
Batch processing generally takes place in some form of agitated equipment, eg a mixing vessel, agitated dryer, but it could also take place in tray dryers.
Non-steady state heat transfer can be illustrated by the following equation. For cooling a batch, the log mean temperature difference can be defined in the following way.
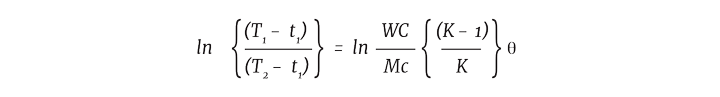
Where
T1 = initial batch temperature
T2 = final batch temperature
t1 = cooling fluid inlet temperature
W = mass flow of cooling media
C = specific heat of cooling fluid
M = mass of the batch in the vessel
c = specific heat of the batch
K = eUA/WC
A = heat transfer area
θ = time
This formula shows that not only is the batch temperature changing over time, so is the impact on other parameters, such as the physical properties of the process materials including the specific heat density, viscosity and conductivity, all of which have an impact on the overall heat transfer coefficient.
The heat transfer area may also not be constant as well. If you heat or cool a liquid, then the density changes and the volume of the liquid changes so that the wetted surface area in the mixing vessel will change to a certain extent. In the case of evaporating the batch, as in batch distillation or concentration, the impact of the reduction in the heat transfer area could be considerably more.
Type of batch heat transfer
At its simplest, batch heat transfer consists of heating or cooling a batch of liquid, liquid/solid mixture or a powdered solid. However, there can be some complexity. Firstly, is there an exothermic or endothermic reaction taking place or not? The other obvious piece of complexity is a change of phase. This could be vaporisation, or it could be crystallisation/solidification. Vaporisation would occur during batch distillation, operation under reflux, concentration or drying. Crystallisation/solidification could occur as part of a crystallisation process or maybe as part of a drown out (a non-solvent is added to cause a material to precipitate), or concentration that leads to precipitation.
The scaleup problem
The scaleup problem can be put simply. The volume of a piece of equipment goes up by cube whereas the surface area only goes up by the square. This means that a process that works at the laboratory scale by simply heating or cooling the outside of the vessel may soon reach a point upon scaleup where the surface area of the vessel is not great enough to allow the required heat transfer to take place.
The scaleup can be solved by several variations on providing additional heat transfer area. Classically this would be a coil inside in the vessel including helical coils (Figure 1b) or serpentine coils that are used to replace the baffles. This approach has two problems, firstly there is a limit to how much additional surface area can be added, and secondly it may be difficult to clean the coils, particularly if solid is deposited on the surface of the coils.
Recent Editions
Catch up on the latest news, views and jobs from The Chemical Engineer. Below are the four latest issues. View a wider selection of the archive from within the Magazine section of this site.




